 |
 |
 |
 |
 |
 |
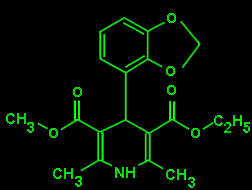 |
OXODIPINE
Calcium antagonist :Oxodipine was developed as a photoresistant, long-lasting, vascular smooth muscle selective calcium channel blocker. Status: Preclinical Pharmacology and Toxicology have been completed Phase I clinical studies have been completed in more tan 200 patients. Phase II studies have been carried out both by intravenous and oral administration in patients with mild to moderate hypertension, demonstrating the efficacy of oxodipine as once-a-day drug. The Oxodipine Project received the Eureka label in 1987 (EU-59) with credits of 219 Million Pesetas from the CEDETI. Oxodipine was discontinued by Synthélabo en 1993 due to compatibility problems with their calcium antagonist Diltiazem. |
