 |
 |
 |
 |
 |
 |
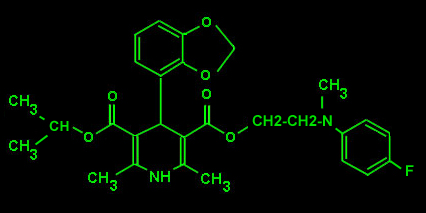 |
ELGODIPINE
Calcium antagonist: Elgodipine was developed on the base of its coronary selectivity and the relative absence of cardiac effects. Status:
Preclinical Pharmacology and toxicology: completed Elgodipine
has been licenced to DELAGRANGE (Synthélabo) and to
ALMIRALL/PRODESFARMA Group. |